
Loading...
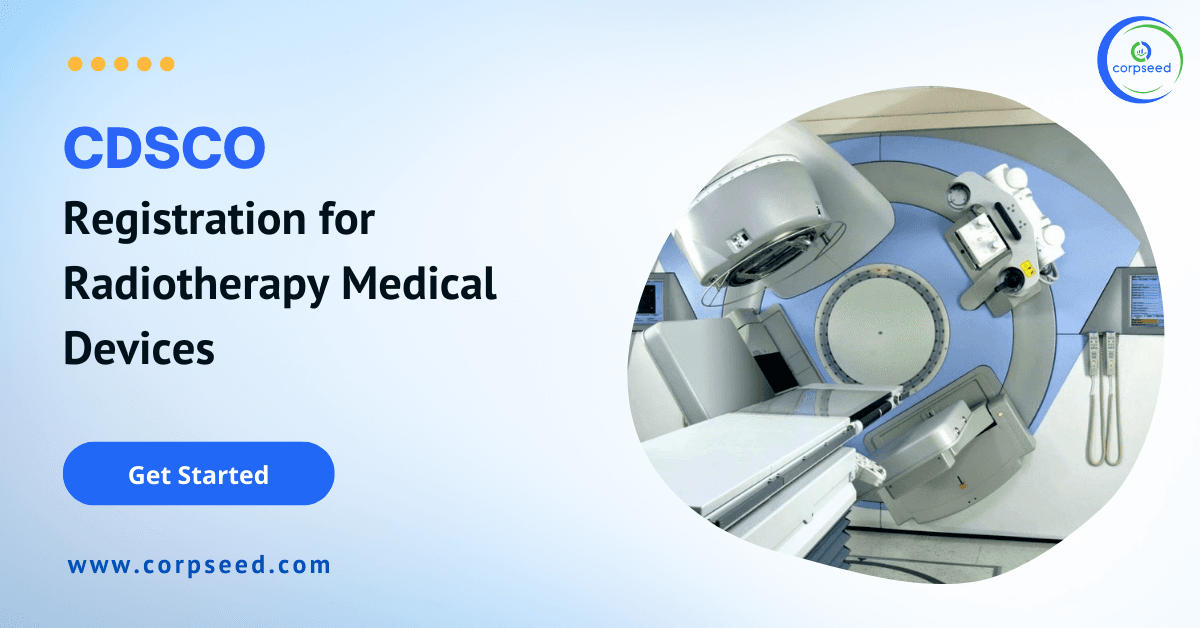
Radiation therapy is used to destroy cancer cells using high–energy x-ray or other particles. A radiation oncologist is a specialist who gives radiation therapy to treat cancer.
About the Author

I have worked for many internet blog pages and news portals. currently, I am working as a content writer for Corpseed Pvt. Ltd. I like to write blogs and articles in the field of different services.
Related articles

CDSCO Drug License Approval in India: Full Process 2026
2026-04-22

CDSCO Mandates ONDLS Portal for r-DNA Drug License Applications
2026-03-16

New Drug Manufacturing and Clinical Trial Rules Updated in 2026: What You Should Know?
2026-03-13

CDSCO Releases New Guidelines for Compounding of Offences Under the Drugs and Cosmetics Act
2026-02-26

Regulation of Medical Device Pricing and Quality in India: Key Government Measures
2026-02-20

CDSCO Releases Risk Classification List for Oncology Medical Devices
2026-02-12
2026-05-05 • 0 views
2023-02-27
2026-05-01